End-to-end Sample Management Solutions
Science requires hundreds or thousands of samples in multiple global locations. That means you may have sample tracking and visibility challenges to remain regulatory compliant.
Avantor® solves the complexities of your sample management needs, from custom kitting to biorepository services.
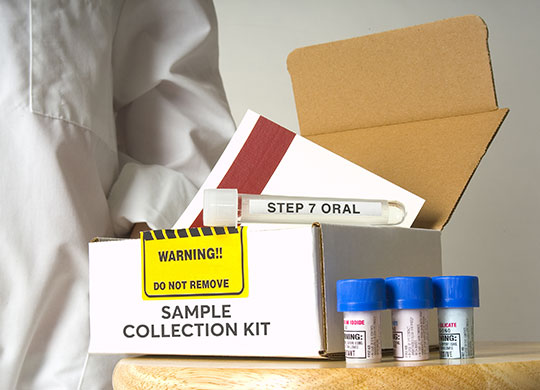
Clinical trial and lab sample management solutions from sample collection to sample storage
You need a trusted partner to provide you confidence in the meticulous care given to your vital research assets, from collection and operations to transport and storage. That means you can focus your time and resources on commercially and scientifically valuable, regulatory-compliant research and patient outcomes.
Benefits of sample management
Our end-to-end sample collection kitting solutions and sample storage biorepository services help you consolidate and save – time, money and resources.
Mitigate risk with our cohesive sample management system
When you partner with Avantor, you can be confident that your vitally important research work will be treated with the same care you would give it.
Benefit from the operational efficiencies of working with a single partner who can integrate its proprietary sample management systems with your CRM and LIMS platforms to ensure full visibility of your kits and samples.
Avoid costly delays in your sample collection workflows
Avantor’s full-service kitting services can advance your pre-clinical, clinical and lab objectives with the agility to deliver reliable, customized sample collection kits. Our tracking and visibility of inventory, plus data-driven insights are features of our kitting services that can help streamline your operations.

Reliable, temperature-controlled sample storage
We offer cold chain transport and biorepository storage services to handle your pre-clinical and clinical assets, with varying complexity and compliance needs. Avantor’s high-quality facilities meet your stringent quality assurance and regulatory objectives for temperature-diverse storage and biorepository services.